What is colloidal silver?
Colloidal silver has been "loosely defined" as a suspension of very small silver particles and silver ions evenly dispersed in a water-based solution.
To be more correct, colloidal silver is properly defined as a solution comprised of silver particles rather than silver ions. Alternately, ionic silver is a solution comprised of silver ions rather than silver particles.
The unit of measure used when referring to these ions and particles is the "nanometer" and this is where we derive the popular term "nanosilver." Keep in mind that "nano" refers only to size and not to a type of silver. This is just a popular marketing term in recent years to imply this to be a completely new type of silver solution which is simply not true.
To understand just how small these particles and ions are, imagine that we magnify the diameter (not length) of a single human hair to represent the distance of one mile. Now, imagine that these tiny silver species would be seen as green peas within that very long one mile stretch.

Watch the Video!
What is nanosilver?
With all of the recent hype about "nanosilver", many may be surprised to find that this is simply the latest marketing term for colloidal silver.

Watch the Video!
What is PPM?
The term "parts per million" or (PPM) is a way of expressing very diluted concentrations of one substance dispersed in another substance.
Silver PPM refers to a quantity rather than a type or size of silver.
Many will confuse 10 PPM and 20 PPM by thinking this is referring to the type or particle size of silver, when the only difference is the total amount of silver. This would be like thinking a 500 mg aspirin is different than a 1000 mg aspirin when the only difference is the quantity of the exact same compound.
1 PPM equates to 1 milligram of silver per 1 liter of water.
For example, if you had a bottle of 500 mg strength aspirin but needed a total dose of 1000 mg, the obvious solution is to ingest (2) of the 500 mg aspirins to achieve the same total dosage.
Let's apply the same analogy to silver by saying you can administer one ounce of a 20 PPM solution or two ounces of a 10 PPM solution.
What kind of silver is produced?
- Silver Ions -
A single silver atom missing one of its negatively charged electrons.
By removing an electron from an electrically neutral silver atom, there is now more positive force within the atom. The (countering) positive force in an atom comes from the fact that there are always an equal number of (+) protons as (-) electrons.
- Silver Atoms -
The smallest individual piece of silver that can exist in its complete state.
Silver atoms will always combine with other silver atoms to form particles of silver. This is called "agglomeration" and these particles of metal are properly termed as "colloids."
- Particles (Colloids) -
Explained previously, particles are two or more silver atoms metallically bonded together.
Particles (colloids) can range in size from ultra-small to considerably large. The smaller a particle is, the better. This is where such close attention to detail and design has been incorporated into The SilverLungs Generator as to ensure only the smallest and highest quality particles are produced.
- Silver Oxide (Ag2O) -
A compound of silver that is formed during the electrolysis process that slowly dissociates over time after the process completes.
This silver compound is formed by two atoms of silver and one atom of free oxygen. Silver oxide will decompose slowly over time to form a diatomic particle of silver and the oxygen is released as free gas.
We simply consider silver oxide to be ionic silver as this is ultimately what final form the silver oxide will take once administered systemically.
Is colloidal silver supposed to be clear or yellow?
The yellow color happens when silver nanoparticles selectively absorb just the purple color spectrum of white light and in-turn the particles reflect back all other colors.
Yellow is the resulting color of white light in the absence of purple and there is simply no way around the yellow color of a true colloidal silver solution.

This phenomenon of physics is called "plasmon absorption resonance" which is not a theory, but rather a very well understood characteristic of silver nanoparticles in solution.
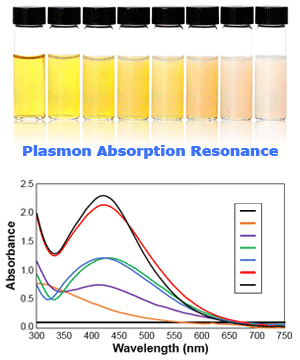
Claims that colloidal silver solutions should always be colorless come from those that are simply unable to produce a "true" colloidal product.
What really happened to "The Blue Man?"
Paul Karason
made the mistake of adding common table salt (sodium chloride) to his distilled water as a catalyst to speed the production time of his homemade silver solutions.Paul was unaware that salt or other additives should never be incorporated into the "electrolysis" process when creating a proper silver solution.

While adding salt reduced Paul's production time, it actually created a completely different silver compound called silver chloride. This compound has been shown to cause a skin discoloration condition called "Argyria." Silver chloride is by no means pure ionic or colloidal silver.
No cases of Argyria have ever been linked to properly produced ionic or colloidal silver solutions.
Does SilverLungs use constant current?
One of the most misreported features of some colloidal silver generators is something marketed as "constant-current." The name is also an absolute misnomer by implying that the current through the water is constant, when in fact this is only true for about 15% of the total time involved in the electrolysis process.
The correct terminology should be "current limiting" as this is the only functional truth to how it works.
The SilverLungs Generator will produce solutions faster than constant current generators while never reaching a critical point in the production that will begin to promote large particulate creation through secondary electro-chemical processes.
The SilverLungs Generator addresses the worry of large particulate creation through its massive 7 gauge silver electrodes, magnetic stirring system, dynamic polarity reversal technology™ and an automatic shut-off feature. All of these features account for keeping the ever-rising current flow below a threshold that would eventually lead to large particulate creation.
What is the shelf-life?
Assuming that your silver solutions are kept in a dark environment and that the storage container you've selected is contamination-free, your solutions will hold in suspension for at least a year and often much longer.
How fast is the generator?
The SilverLungs Generator produces a 10 PPM solution in 90 minutes and a 20 PPM solution in 150 minutes.
How long do the silver electrodes last?
The original SilverLungs electrodes should last almost indefinitely even with constant and extended use.
Always remember to use the provided copper polishing pad to ensure that the electrodes are polished rather than ground down when using the highly-abrasive "Scotch Brite" type scrubbers that come with other generators.
Scotch Brite scrubbers are extremely abrasive and remove far more silver during the scrubbing process than during silver production.
This is because copper carries a very similar hardness to silver and makes it the optimal material of choice for polishing (rather than grinding) them.
Are the electrodes pure?
The SilverLungs Generator utilizes electrodes based on 99.99% pure elemental silver. The 4th "9" of purity ensures that you are producing the highest quality solutions possible.
Each SilverLungs electrode comes polished and straightened with no sharp edges ensuring an even electrical flow between the two.
The large diameter and length of the silver electrodes (combined with a very large silver-to-water contact area) allows for a near-infinite life-span.
Silver Purity Certification
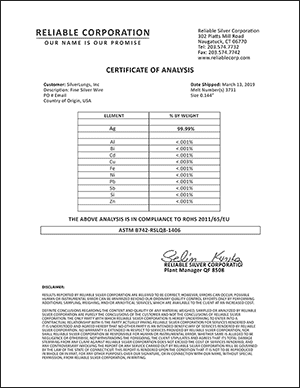
click image to view
Why use a laser pointer?
A high-powered red laser pointer is an indispensable tool for checking the quality of your silver solutions.
The presence of large and undesirable silver complexes will reveal themselves easily with a red laser pointer.
While The SilverLungs Generator has been carefully engineered to create clean and consistent silver solutions that are free of large and undesirable silver complexes, there is still nothing more reassuring than a quick visual inspection of your product.
What is magnetic stirring?
The SilverLungs Generator features a magnetic stirring system that does not require the flask to be un-sealed during silver production. Many competing silver generators do not even include a stirring mechanism, while the ones that do are not the sealed type. This leaves the solution exposed to air-born contaminants.
Other stirring methods like "bubblers" are very turbulent and tend to loosen the excess silver hydroxide build-up which inherently collects. This build-up gets deposited into the final solution and can creates a messy end-result.
Dynamic polarity reversal?
Standard polarity reversal is already a very useful and functional feature in a silver generator as it spreads the wear and load of the dissolving silver evenly across both electrodes.
Dynamic polarity reversal technology™ uses very specific and ever-changing timing intervals as an advancement to standard "fixed timing" methods. This greatly reduces the total amount of silver hydroxide that inherently collects on the electrodes during production.
Why use a water tester?
Our digital water test meter is vital for testing the initial quality of your production water before you begin and also allows for accurate measurement of the total amount of silver dissolved.
The water test meter comes pre-calibrated specifically for silver.
Why only distilled water?
Water that is not properly distilled or deionized is essentially not pure and may contain undesirable elements that can interfere with the final quality and effectiveness of a new silver solution.
It is also imperative that no "additives" be incorporated in the electrolysis process to ensure that only pure silver is present within pure water.